A GMP warehouse plays a critical role in ensuring products are stored, handled, and distributed in compliance with strict regulatory and quality requirements.
GMP standards help protect product integrity throughout the supply chain. Businesses operating in regulated industries must meet these expectations to maintain compliance, reduce risk, and ensure consumer safety.
This guide explains the core requirements and best practices that help organizations operate a GMP-compliant warehouse with confidence.
Key GMP Regulations for Warehouses
While there are several GMP regulations in force, we wanted to discuss some of the most important ones that apply specifically to warehouse operations.
These GMP warehouse center requirements focus on protecting product integrity, preventing contamination, and maintaining consistent quality throughout storage and distribution.
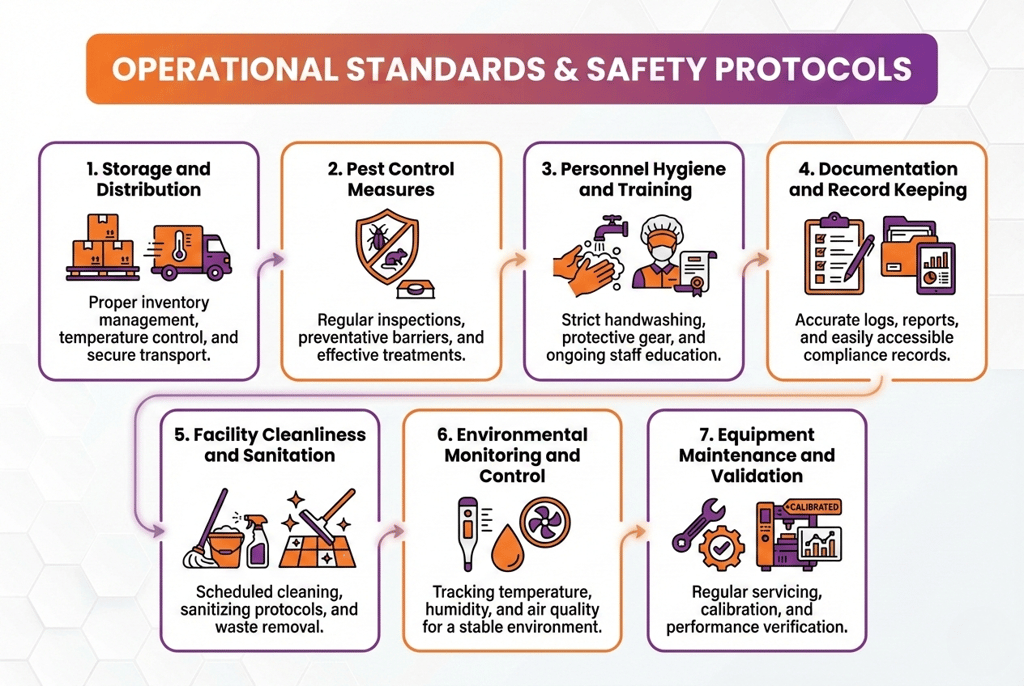
1. Storage and Distribution
Proper storage and distribution practices are at the core of GMP warehouse rules. Companies looking to enhance efficiency can explore inventory management solutions to ensure product integrity and minimize errors. This includes:
- Maintaining suitable temperature and humidity conditions
- Organizing storage areas to prevent cross-contamination
- Implementing efficient inventory management systems to minimize errors and product mix-ups
Furthermore, a GMP warehouse must have designated storage areas for different types of products based on their specific requirements.
For example, storing temperature-sensitive items should be done in climate-controlled zones to maintain their integrity and efficacy. Proper labeling and segregation of products are also essential to avoid mix-ups and ensure accurate tracking of inventory levels.
2. Pest Control Measures
Pests have no place in a GMP warehouse, and GMP rules emphasize the implementation of robust pest control measures. This involves:
- Regular inspection
- Proper sanitation
- The use of appropriate pest control products to prevent infestations
In addition to routine pest control measures, a GMP warehouse should also focus on identifying and addressing potential entry points for pests.
This includes sealing cracks and crevices, maintaining cleanliness both inside and outside the facility, and implementing monitoring systems to detect any signs of pest activity promptly.
By taking a proactive approach to pest control, warehouses can safeguard their products and maintain compliance with GMP regulations.
3. Personnel Hygiene and Training
Human factors play a critical role in product quality, making personnel hygiene and training a core requirement of GMP warehouse rules. Employees must follow strict hygiene practices to reduce contamination risks, while training ensures they understand and comply with GMP standards.
Key hygiene and training measures include:
- Proper handwashing and use of appropriate protective clothing
- Clear hygiene protocols for handling and storage activities
- Regular GMP training to reinforce compliance responsibilities
- Periodic audits to assess hygiene practices and identify improvements
By promoting consistent hygiene standards and ongoing training, warehouses reduce human error, maintain compliance, and protect product integrity throughout operations.
4. Documentation and Record Keeping
Accurate documentation is a core requirement of GMP-compliant warehousing operations. Records provide traceability, accountability, and evidence that procedures are being followed correctly.
This typically includes documentation related to:
- Inventory movements and stock levels
- Storage conditions and environmental monitoring
- Cleaning, sanitation, and pest control activities
- Employee training and compliance records
Clear, well-maintained records support audit readiness and make it easier to investigate issues, manage deviations, and demonstrate regulatory compliance.
5. Facility Cleanliness and Sanitation
GMP regulations require warehouses to maintain clean, sanitary environments to prevent contamination and protect product integrity. Cleaning procedures must be clearly defined, documented, and consistently applied across all storage and handling areas.
Key sanitation requirements include:
- Scheduled cleaning routines for all warehouse zones
- Use of approved cleaning agents suitable for stored products
- Documented cleaning procedures and records
- Proper waste management to prevent contamination
Maintaining high sanitation standards supports GMP compliance and ensures a controlled storage environment that protects product quality.
6. Environmental Monitoring and Control
Maintaining appropriate environmental conditions is essential for GMP-compliant warehousing, particularly for temperature- or humidity-sensitive products.
Environmental control requirements for GMP warehouses typically include:
- Monitoring temperature and humidity levels
- Using calibrated sensors and monitoring equipment
- Setting alerts and corrective actions for deviations
- Maintaining records of environmental data over time
Effective monitoring helps warehouses identify risks early, prevent product degradation, and maintain consistent compliance with GMP storage requirements.
7. Equipment Maintenance and Validation
Warehouse equipment used for storage, handling, and monitoring must be properly maintained and validated to meet GMP standards. This includes racking systems, material handling equipment, and environmental monitoring devices.
Key equipment maintenance requirements include:
- Scheduled maintenance and servicing of warehouse equipment
- Regular calibration of monitoring and measuring devices
- Documented inspections and validation records
- Prompt repair or replacement of faulty equipment
Proper equipment maintenance reduces operational risk and supports consistent GMP compliance across warehouse operations.
GMP Rules for a Warehouse 101: Understanding Compliance

GMP, or Good Manufacturing Practices, is a set of principles and guidelines established by regulatory agencies such as the U.S. Food and Drug Administration (FDA).
These rules are tailored to different industries, outlining the procedures and protocols necessary to maintain product quality and safety throughout the supply chain.
GMP Warehouse Meaning
A GMP warehouse meaning refers to a storage and distribution facility that operates in full alignment with Good Manufacturing Practice standards.
This means the warehouse is designed and managed to protect product integrity through controlled environments, documented processes, trained personnel, and strict hygiene protocols.
A GMP-compliant warehousing ensures that products are stored, handled, and moved in a manner that prevents contamination, reduces risk, and maintains quality from receipt through dispatch.
This level of control is especially crucial for regulated industries, including pharmaceuticals, food, supplements, and medical devices.
Importance of GMP in Warehousing
Warehouses play a crucial role in the supply chain, storing and distributing products before they reach end customers.
Adhering to GMP rules in warehousing ensures products are handled and stored in a way that minimizes contamination risks while preserving quality, safety, and efficacy.
Implementing GMP standards in warehouse operations typically involves:
- Maintaining proper storage conditions, including controlled temperature and humidity
- Preventing contamination through clean layouts and defined handling procedures
- Training staff on GMP protocols and hygiene requirements
- Documenting processes to support traceability and audit readiness
By applying GMP principles consistently, warehouses reduce operational risks, support regulatory compliance, and maintain the integrity of products throughout storage and distribution.
Implementing GMP in Your Warehouse Operations
Good Manufacturing Practices (GMP) play a vital role in ensuring the quality and safety of products in warehouse operations.
A GMP-compliant warehousing follows structured processes that support product integrity, regulatory compliance, and consistent handling standards.
Developing a GMP program tailored to the specific needs of your warehouse is essential for maintaining high standards and meeting regulatory expectations.
In addition to outlining standard operating procedures, a robust GMP program should also encompass training programs for staff to ensure understanding and adherence to GMP guidelines.
Developing a GMP Program
To become a proper GMP warehouse, the facility must develop a comprehensive program tailored to its specific operations. A strong warehouse GMP framework ensures that procedures are applied consistently across daily activities.
This program should:
- Outline standard operating procedures
- Define responsibilities and accountabilities
- Establish a system for ongoing monitoring and continuous improvement
Regular training sessions and refresher courses reinforce GMP expectations among warehouse staff and help foster a culture of quality, consistency, and accountability.
Regular Audits and Inspections
A crucial aspect of GMP implementation in warehouses is conducting regular audits and inspections. These assessments help identify gaps in compliance, verify adherence to GMP principles, and highlight opportunities for corrective actions and process optimization.
Third-party audits can provide an unbiased evaluation of warehouse performance and offer valuable insights for improvement.
Implementing a robust internal auditing system also helps warehouses remain proactive in addressing issues and continuously improving operational standards.
Challenges in Adhering to GMP Rules Effectively

Ensuring compliance with Good Manufacturing Practices is critical for maintaining product quality and safety, but it can present operational challenges. Applying GMP in a warehouse environment requires discipline, documentation, and ongoing staff engagement.
Warehouses commonly face the following challenges:
- Inadequate employee training, where staff lack sufficient knowledge of GMP requirements
- Incomplete documentation, which weakens traceability and audit readiness
- inconsistent implementation of standard operating procedures across teams or shifts
Addressing these issues requires a structured, proactive approach and a commitment to continuous improvement, but ultimately leads to an efficient and functioning GMP warehouse.
Common Compliance Issues
Despite their importance, GMP rules can be difficult to enforce consistently. Inadequate training, incomplete records, and procedural inconsistencies are frequent causes of non-compliance.
Identifying these issues early helps prevent escalation and supports long-term operational stability.
Overcoming GMP Implementation Obstacles
Overcoming GMP challenges starts with identifying the root causes of non-compliance. By conducting thorough assessments and engaging employees at all levels, warehouses can address obstacles such as:
- Resistance to change
- Limited resources
- Organizational cultures that do not prioritize quality
Fostering open communication and continuous learning helps align teams with GMP objectives and strengthens compliance across operations.
The Road to a Compliant GMP Warehouse
When warehouses implement GMP best practices, they not only ensure compliance with regulations but also set a benchmark for operational excellence.
This includes regular training sessions for employees on GMP principles, conducting thorough inspections of storage areas, and maintaining detailed records of all processes.
By investing in these practices, GMP warehouses can proactively identify and address any potential issues before they escalate, thus maintaining a high level of quality and efficiency.
For businesses looking to optimize their fulfillment and logistics operations, Fulfyld provides expert solutions to streamline processes and enhance efficiency.